Had to walk down a lley with alk ies ly ing down.The angel presented the thief she had caught to the mayor. Halogens What happened next was incredible! An angel swooped down and caught one of the thieves by snaring him with her halo! ( Halogens) Chalcogens I could see the poor store owner inside, adding up what had been taken on his calculator. Pnictogens But, as I watched, thieves suddenly ran up and smashed the glass! The necklace quickly got nicked! ( Pnictogens)ħ. Crystallogens The most impressive thing they were selling was a glittering necklace made of crystal. ( Crystallogens)Ħ. 
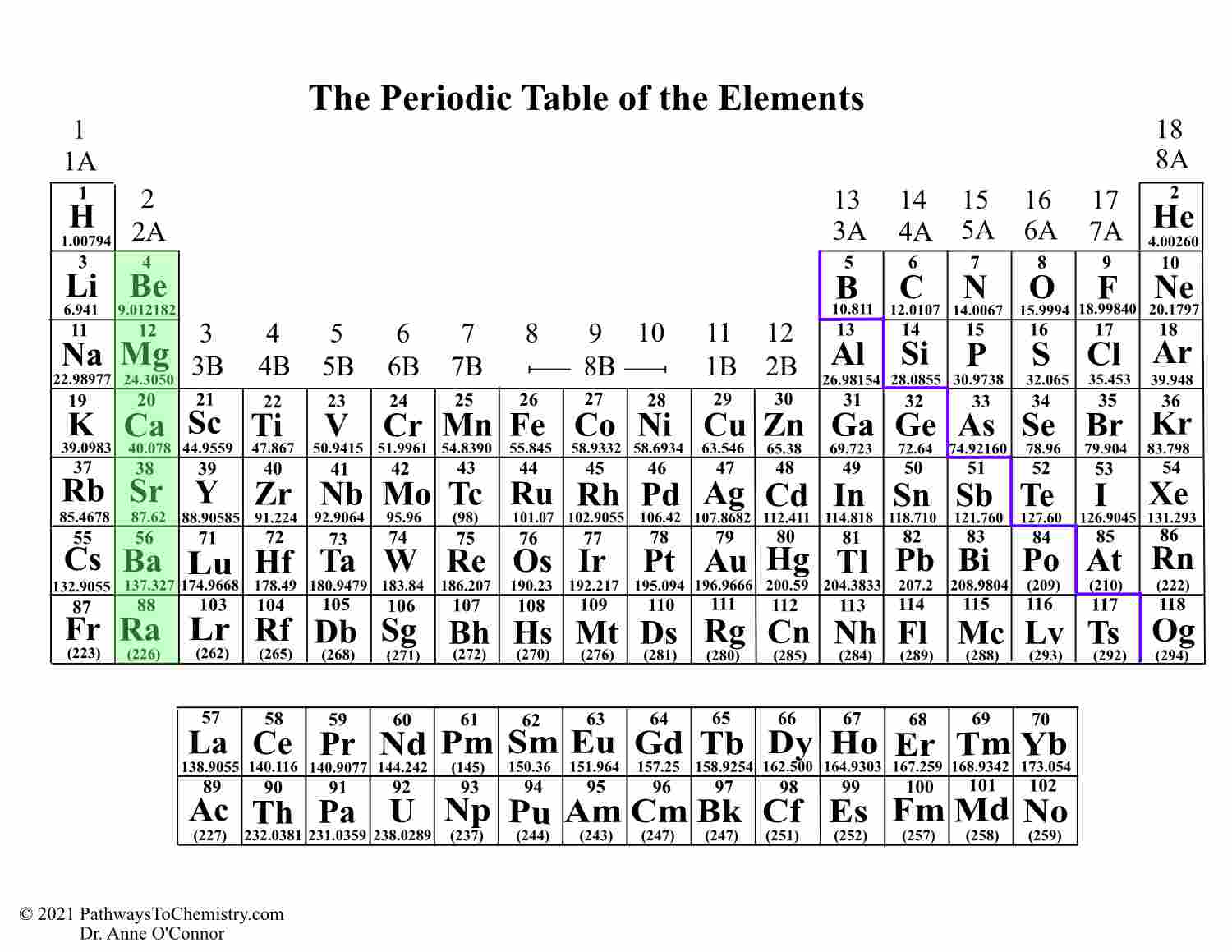
We drove safely out of the alley and a very expensive jewellery store caught my eye – it was selling high cost gems. ( Icosagens)ĥ. Transition metals We were so worried about walking that we drove through instead, in a transit van. The word alkaline refers to something entirely different. Never call this group just the alkalines these elements should always be referred to as the alkaline earth metals. Alkaline earth metals Looking down, I saw that the street itself was paved with bottles and cans of alcohol in line s in the earth. Alkalis on their own are something entirely different. Never call this group just alkalis, it should always be referred to as the alkali metals. Alkali metals We were all very nervous because we had to go down a back street full of drunks, all the alkies were lying down with their metal beer cans.( Alkali metals) We were talking about groups, which is a coincidence – I was out for a walk with a group of friends just the other day. To remember the names of the groups, use the following story: The groups most discussed in school Chemistry are: They have some pretty weird names, though, which are very difficult to remember… unless you use the Mammoth Memory story technique, of course! All of the elements in a group behave in the same way. We created a timeline of the history of the periodic table.All of the elements in the periodic table are divided up into groups. Key groups include alkali metals, alkaline earth metals, halogens. The table is divided into metals, nonmetals, and metalloids, each with distinct properties. Elements in the same group share similar characteristics, like reactivity. The Periodic Table has constantly been improved and developed over the past 200 years, but in 1869 Dimitri Mendeleev finished the first version of the periodic table as we know it today, by arranging the elements by atomic mass and leaving spaces open for the elements that were not yet discovered. The periodic table organizes elements into groups and periods based on their chemical and physical properties. Who invented the Periodic Table of Elements? The periodic table also gives us an idea of what the characteristics of an element might be and help us predict how an element might react based on in which group it is located. The Periodic Table of Elements can be used as an assisting tool in chemical calculations, when a specification of an element is needed it is easily found in the Periodic Table. How is the Periodic Table of Elements used? The table lists all the elements that are currently known (118), in descending order of the number of protons that are present, in a single atom of the element. For each of the following elements, write its chemical symbol, determine the name of the group to which it belongs (Table 2.3), and indicate whether it is a metal, metalloid, or nonmetal: (e) bromine. The Periodic table of elements is a tool, developed by scientists over hundreds of years. Identify the three elements indicated on the periodic table, and give the group that they are in. List of all the elements and their properties:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
